According to the National Institute of Diabetes and Digestive and Kidney Diseases: "Normal, healthy kidneys filter about 200 quarts of blood each day, generating about 2 quarts of excess fluid, salts, and waste products that are excreted as urine. Loss of function of these organs, even for a short period of time or due to gradual deterioration, can result in life-threatening complications. Whether kidney function is lost suddenly or slowly represents an important health challenge.
It has been estimated that more than 30 million American adults have chronic kidney disease (CKD). CKD has two main causes: high blood pressure and diabetes. CKD, especially if undetected, can progress to irreversible kidney failure."

Michael Brands, PhD - Regents' Professor - Physiology
706-721-9785 / mbrands@augusta.edu / CV
Cardiovascular-renal integrative physiology and hypertension. Longstanding interest in renal and hormonal mechanisms for chronic blood pressure and circulatory system control in states of insulin resistance, hyperinsulinemia, and diabetes.

Mykola Mamenko, PhD - Assistant Professor - Physiology
706-721-9103 / mmamenko@augusta.edu / CV
Our research is focused on the cellular and molecular mechanisms governing water and electrolyte handling in the kidney. The laboratory pursues a few lines of inquiry: we study how disrupted calcium homeostasis in renal epithelial cells is associated with polycystic kidney disease (PKD) and nephrogenic diabetes insipidus (NDI), investigate sex-specific mechanisms governing final regulation of sodium reabsorption in the kidney, critical for the chronic blood pressure control.

David Mattson, PhD - Department Chair and Professor
706-721-7742 / dmattson@augusta.edu / CV
Studies in the Mattson laboratory examine the normal and pathophysiological regulation of renal function and arterial blood pressure. A particular emphasis is placed on the paracrine, autocrine, and hormonal regulation of renal tubular and vascular function. Additional studies are geared toward an understanding of the genetic basis of hypertension and renal disease.

Paul O'Connor, PhD - Associate Professor - Physiology
706-721-7890 / paoconnor@augusta.edu / CV
Our laboratory’s primary research interests lie in the physiological pathways involved in the regulation of kidney function and how disruptions in these pathways can lead to disease. Recently our laboratory has also become interested in the mechanisms through which splenic anti-inflammatory pathways regulate the innate immune response.

Jennifer Sullivan, PhD - Professor - Physiology
706-721-9796 / jensullivan@augusta.edu / CV
The overall goal of my laboratory is to better understand the molecular mechanisms that regulate blood pressure in males and females under both physiological and pathophysiological conditions, including hypertension. Traditionally, it has been assumed that blood pressure control and the basis of hypertension is the same in males and females; just the magnitude of the response differs. However, based on the vast number of differences that have been identified in cardiovascular physiology, pathophysiology, and pharmacology between the sexes, there is growing evidence to suggest that the pathways by which males and females develop cardiovascular and renal diseases may be distinct. Ongoing studies are focused on 3 pathways involved in blood pressure control and cardiovascular function: the renin angiotensin system (RAS), the nitric oxide (NO) pathway, and inflammation.
"Scientists have become the bearers of the torch of discovery in our quest for knowledge." - Stephen Hawking
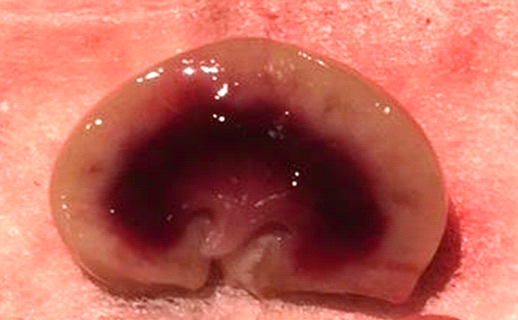
Research image from Dr. O'Connor's lab showing vascular congestion (dark red) in the renal outer-medulla of the rat kidney following a period of ischemia
